
Capital Raising
CLOSED
Oscar Resources Limited
Iron Ore company utilising breakthrough technology
INDUSTRY
Medical & Technology
This offer closed November 2022. If you would like to discuss anything in relation to this company please contact us for further information.
All information on this page was current as at the date of closure.
Investment Highlights
Biosignal monitoring company
Prevent, diagnose, and change outcomes for heart disease & stroke
ECGme device provides cost effective & real time solutions
Precision Healthcare
Company Overview
Products & Services
Biosignals Diagnostics offers solutions that target unmet clinical needs or improved standard of care in heart disease, with verified customers and markets.
Biosignals Diagnostics is a biosignal monitoring company, that addresses heart disease problems by integrating biometric data from specially designed, easily used devices and other sources such as imaging and genetics.
Product Suite
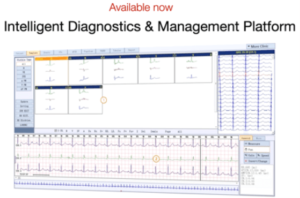

ECGME
- Small easy to use the device (the size of a credit card), fits in your pocket and when held with fingers on the sensors analyses real-time ECG signals
- Point-in-time ECG snapshot configurable for up to 10 minutes monitoring duration
- Converts to a 2 electrode 24hr on-body monitor for clinical use
- A TGA-approved Class 2a medical device
- Available July 2022

Want to learn more?
Fill in the expression of interest form below

